Recognized as revolutionary by Chemical & Engineering News, Colorado State University has found a new chemical tool for synthesizing important molecules.
CSU chemistry professors Andrew McNally and Robert Paton have discovered a new, straightforward reaction for applications in synthesizing drugs. With the help of chemistry Ph.D. students Michael Hilton and Benjamin Boyle and postdoctoral research associates Juan Alegre-Requena and Xuan Zhang, the two labs have combined research from the lab and research behind a computer to fully create and understand this reaction.
“It’s a really valuable collaboration with people at the bench and people at the screen,” Paton said. “This is a completely new transformation, and nobody knew how it worked, and that’s why we do science. Hopefully, we’ve managed to scratch some of the surface, and I’m sure there’s going to be more things to learn, but we’re at the source. The discoveries are coming out of this corridor and we can get to calculate these things before everybody else.”
McNally and Paton’s labs are both interested in pharmaceutical applications because of the potential impact of medicines on human health. Paton said that modern medicine, which has saved a lot of lives, is fundamentally based in the chemicals both labs work to understand.
“People can often take it for granted that there are cures for diseases and that is fundamentally based on the chemical reactions required for drug development,” McNally said. “The science involved with developing new therapeutics is literally life and death critical.”
If you’ve got something complicated, it’s always going to be easier when you know how it works to do something with it. Otherwise, you’re like a chimp with a wrench.” – Andrew McNally, chemistry professor
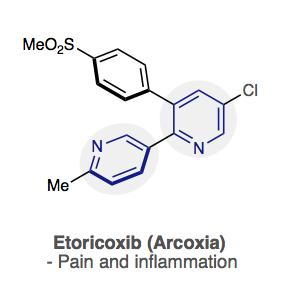
Instead of using a palladium catalyst, which is currently the most common catalyst used to make drugs, the research team got rid of the metal altogether and used phosphorus connected to a pyridine molecule to connect two pyridine molecules together.
Pyridine molecules, a molecule shaped bonded like a ring with five carbons and one nitrogen atom, are especially difficult to bond and to work with because the nitrogen can cause the catalyzed reaction to fail, Boyle said. The reactivity around the phosphorous attached to a pyridine was known but was never applied until this reaction.
Ironically, the nitrogen in the structure is also what makes the pyridine useful because it interacts well with the drug target, which can be a critical part of the mode of action, Paton said.
“We can use this knowledge to design different reactions,” McNally said. “We’ve got a number of new transformations that were entirely born out of the model developed through computation. If you’ve got something complicated, it’s always going to be easier when you know how it works to do something with it. Otherwise, you’re like a chimp with a wrench.”
A unique aspect of this project is the use of computational organic chemistry in order to further understand why the reaction works. Paton, who became a professor at CSU in January 2018, is a part of a newer and growing field that focuses on the computational side of organic chemistry. Computational chemistry allows experimental chemists to “shine a light in a dark box,” by breaking down the mechanism visually and by components, Paton said.
“You can write down a mechanism on the blackboard, and you may think, ‘this is the mechanism, I’m going to try and improve it,’ but you’re very limited experimentally,” Alegre-Requena said. “When you have the mechanism and when you can see it in the computer, you can see these bonds are breaking, or ‘this is what is going on, this group is more important than this one,’ that is what is going to allow you to actually make a huge improvement.”
McNally said because the labs have a good understanding of how the reaction works, the reaction is simple and the reaction is versatile, this has the potential to be used to synthesize new drugs in the pharmaceutical industry.
“To get people to use your chemistry, you need to make it simple, so basically anyone can go into their lab and pretty much immediately apply this,” McNally said. “That isn’t true for a lot of chemistry. Often there are barriers to actually apply it in a different lab outside of where it was developed, but with this, there isn’t really any barriers to applying this science anywhere.”
Collegian reporter Julia Trowbridge can be reached at news@collegian.com or on twitter @chapin_jules.